To coax human nerve cells in a laboratory to thrive, there are three magic words: location, location, location.
Many experiments grow human nerve cells in lab dishes. But a new study enlists some real estate that’s a bit more unconventional: the brain of a rat. Implanted clusters of human neurons grow bigger and more complex than their cohorts grown in dishes, researchers report online October 12 in Nature.
Not only that, but the human cells also appear functional, albeit in very limited ways. The implanted human cells can both receive signals from rat cells and influence the rats’ behavior, connections that “demonstrate more substantial integration of the transplanted neurons,” says Arnold Kriegstein, a developmental neuroscientist at the University of California, San Francisco, who wasn’t involved in the study. “This is a significant advance.”

Sign Up For the Latest from Science News
Headlines and summaries of the latest Science News articles, delivered to your inbox
Thank you for signing up!
There was a problem signing you up.
Over the last decade, scientists have been building increasingly complex brain organoids, 3-D clusters of cells derived from stem cells that grow and mimic the human brain (SN: 2/20/18). These organoids don’t re-create the full complexity of human neurons that develop in an actual brain. But they can be windows into an otherwise inscrutable process — human brain development, and how it can go awry (SN: 9/3/21). “Even if they’re not entirely perfect, [these models] are surrogates for human cells in a way that animal cells are not,” Kriegstein says. “And that’s really exciting.”
To push these cells closer to their full potential, Sergiu Pasca, a neuroscientist at the Stanford School of Medicine, and colleagues surgically implanted human cerebral organoids into the brains of newborn rat pups. Along with their hosts, the human organoids began to grow. Three months later, the organoids were about nine times their starting volume, ultimately making up about a third of one side of the rat’s cortex, the outer layer of the brain. “It pushes the rat cells aside,” Pasca says. “It grows as a unit.”
These human cells flourished because rats’ brains offer perks that lab dishes can’t, such as blood supply, a precise mix of nutrients and stimulation from nearby cells. This environmental support coaxed individual human neurons to grow bigger — six times as large by one measure — than the same sort of cells grown in dishes. Cells grown in the rat brains were also more complex, with more elaborate branching patterns and more cell connections called synapses.
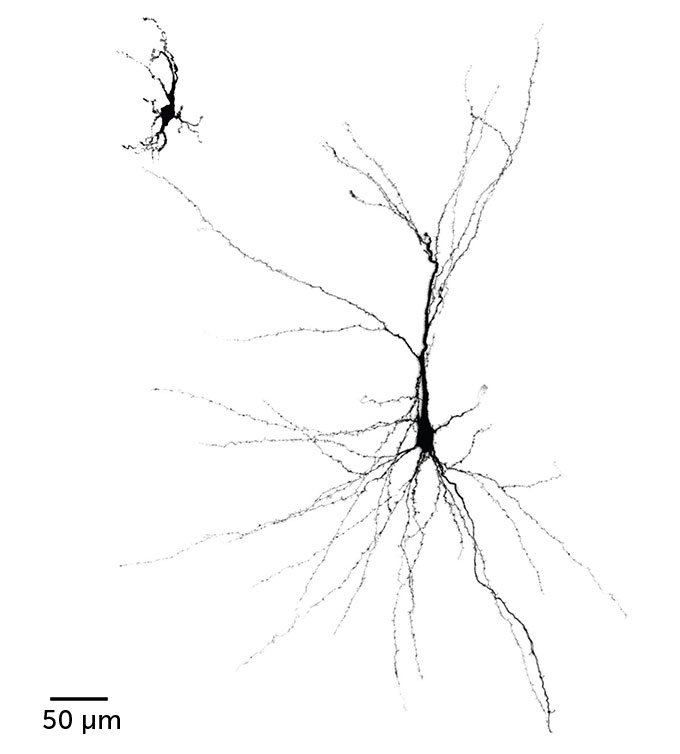 A human nerve cell from an organoid that resided in the brain of a rat (right) grew bigger and more complex than a similar cell grown in an organoid in a laboratory dish (left).Stanford University
A human nerve cell from an organoid that resided in the brain of a rat (right) grew bigger and more complex than a similar cell grown in an organoid in a laboratory dish (left).Stanford University
The cells looked more mature, but Pasca and his colleagues wanted to know if the neurons would behave that way, too. Tests of electrical properties showed that implanted neurons behaved more similarly to cells that develop in human brains than cells grown in dishes.
Over months of growth, these human neurons made connections with their rat host cells. The human organoids were implanted in the somatosensory cortex, a part of the rat brain that handles whisker input. When researchers puffed air at the whiskers, some of the human cells responded.
What’s more, the human cells could influence the behavior of the rat. In further experiments, the researchers genetically tweaked the organoids to respond to blue light. Prompted by a flash of light, the neurons fired signals, and researchers rewarded the rats with water. Soon, the rats learned to move to the water spout when their human organoid cells sent signals.
In behavioral tests, rats with human implants didn’t show signs of higher intelligence or memory; in fact, researchers were more concerned with deficits. The human organoids were nudging out their hosts’ brains, after all. “Will there be memory deficits? Will there be motor deficits? Will there be seizures?” Pasca asked. But after extensive tests, including behavior tests, EEGs and MRIs, “we could not find differences,” Pasca says.
Other experiments included nerve cells from people with a genetic disorder called Timothy syndrome, a severe developmental disorder that affects brain growth. Growing organoids created with these patients’ cells in rats’ brains might reveal differences that other techniques would not, the researchers reasoned. Sure enough, neurons in these organoids had less complex message-receiving dendrites than those from organoids derived from people without the syndrome.
Organoids made from patient-specific cells could one day even serve as test subjects for treatments, Pasca says. “Challenging disorders will require bold approaches,” he says. “We will need to build human models that recapitulate more aspects of the human brain to study these uniquely human conditions.”


